|
In a neutral atom, the number of electrons equals the number of protons. Each such orbital can be occupied by a maximum of two electrons, each with its own projection of spin m s. The chemistry of each element is determined by its number of protons and electrons. As an alternative to the magnetic quantum number, the orbitals are often labeled by the associated harmonic polynomials (e.g., xy, x 2 − y 2). They are assorted in a tabular arrangement wherein a row is a period and a column is a group. Įach orbital in an atom is characterized by a set of values of the three quantum numbers n, ℓ, and m l, which respectively correspond to the electron's energy, its angular momentum, and an angular momentum vector component ( magnetic quantum number). The periodic table is an arrangement of all the elements known to man in accordance with their increasing atomic number and recurring chemical properties. When the periodic table is divided in this. In older IUPAC group numbering systems, the main group elements are groups IA, IIA, and IIIA to VIIIA. These are elements in group 1 and group 2 (s-block) and groups 13 through 18 (p-block). The term atomic orbital may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital. The main group elements are the chemical elements belonging to the s-block and p-block on the periodic table. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. Metals are otherwise defined according to their position on the Periodic Table. In atomic theory and quantum mechanics, an atomic orbital ( / ˈ ɔːr b ɪ t ə l/) is a function describing the location and wave-like behavior of an electron in an atom. The definition of metal: A substance with high electrical conductivity, luster, and malleability, which readily loses electrons to form positive ions ( cations ). To see the elongated shape of ψ( x, y, z) 2 functions that show probability density more directly, see pictures of d-orbitals below. The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. 1: The Periodic Table Showing the Elements in Order of Increasing Z. Each picture is domain coloring of a ψ( x, y, z) function which depends on the coordinates of one electron. The chemistry of each element is determined by its number of protons and electrons. The two colors show the phase or sign of the wave function in each region.  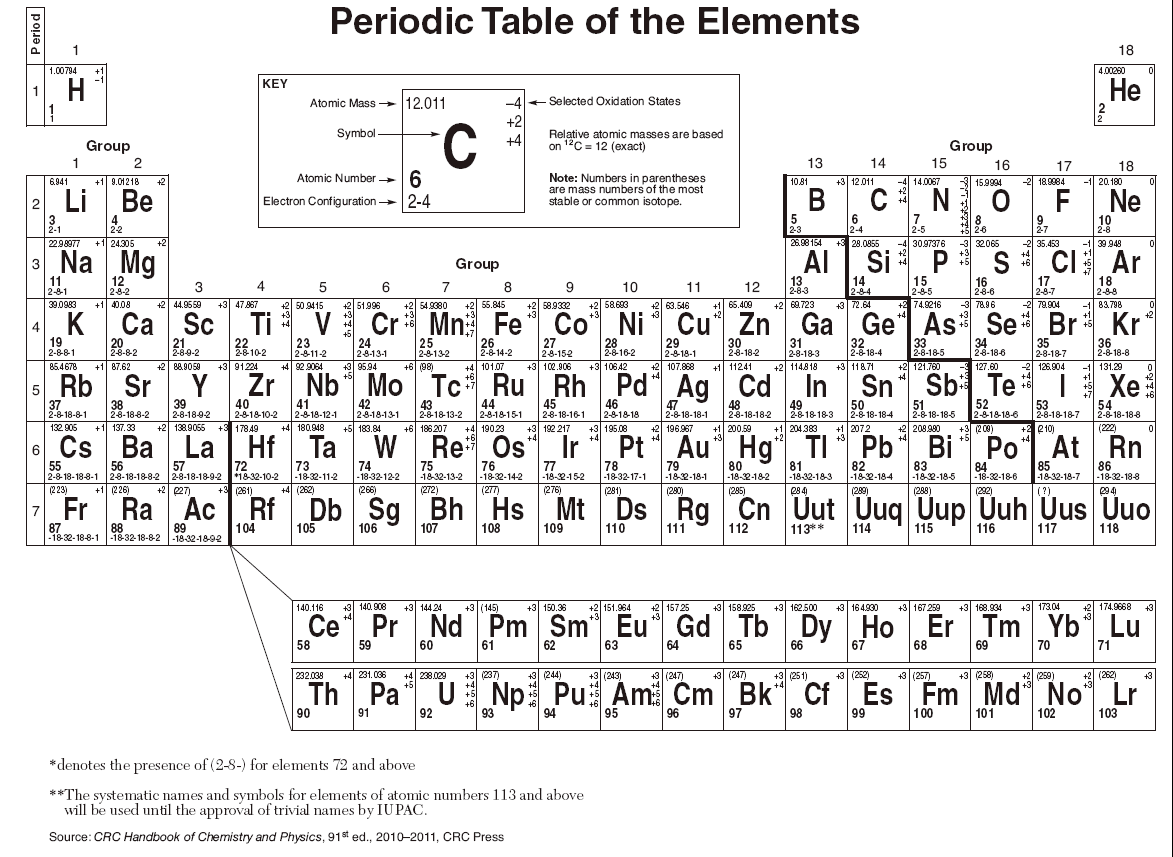
The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z. For the collection of spaceflight orbits, see Orbital shell (spaceflight).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
